Researchers Uncover Glial Cells Metabolic and Inflammatory Roles
A recent study has shed light on the evolving understanding of glial cells in the context of neurodegenerative diseases. Traditionally, the focus has been on neurons when examining disorders such as Alzheimer's disease, Parkinson's disease, amyotrophic lateral sclerosis, ischemic stroke, and hypoxic-ischemic brain injury. However, new research indicates that the progression of these conditions is significantly influenced by the behavior of glial cells, particularly microglia and astrocytes.
Published in Ageing Research Reviews, the study, conducted by a team led by MA Yinzhong at the Shenzhen Institutes of Advanced Technology of the Chinese Academy of Sciences, delves into the role of glial cells in disease progression. It explores how these cells undergo metabolic reprogramming, amplify inflammation, and contribute to neurovascular dysfunction.
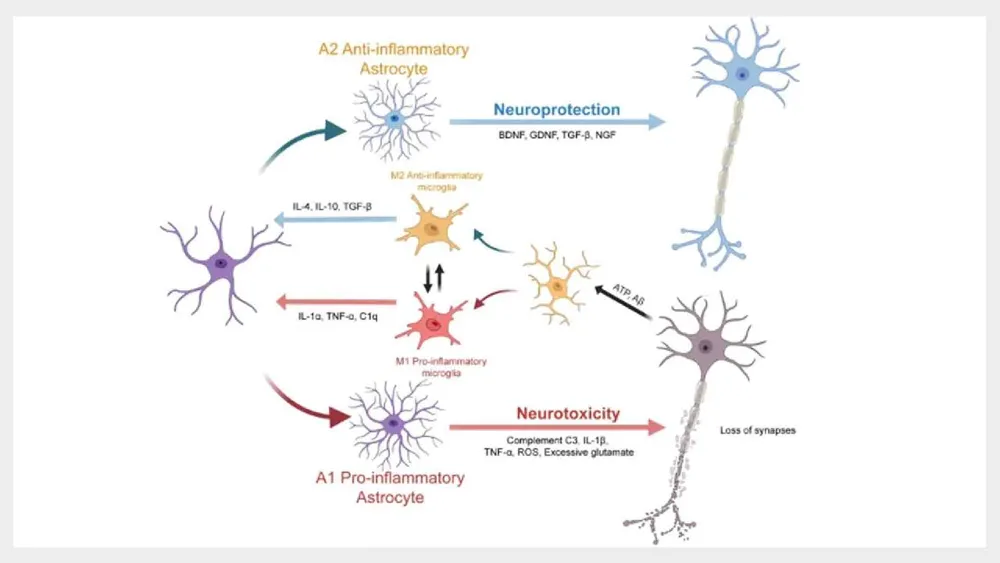
Under normal physiological conditions, microglia and astrocytes are essential for maintaining homeostasis within the central nervous system. Microglia act as vigilant monitors of the brain environment, engaged in immune regulation and debris removal, while astrocytes play vital roles in neurotransmitter recycling, lipid transport, providing metabolic support, and ensuring the integrity of the blood-brain barrier.
In contrast, during disease states, the researchers observed significant functional shifts in these glial cell types. Microglia move from a state of monitoring to one that is characterized by inflammatory responses or repair processes, whereas astrocytes change into reactive phenotypes that can either provide neuroprotection or contribute to neurotoxicity.
The findings further highlight that glial dysfunction is intricately linked to metabolic processes. When activated, microglia often switch from oxidative phosphorylation to glycolysis, resulting in increased production of cytokines and reactive oxygen species. Similarly, astrocytes frequently suffer disruptions in lipid metabolism, leading to impaired cholesterol transport and the accumulation of lipid droplets, which diminishes their supportive functions for neurons and exacerbates inflammatory responses.
Additionally, the study revealed the existence of pathological feedback loops between microglia and astrocytes. Once activated, these cells can amplify each other's effects through various inflammatory mediators, complement signaling pathways, metabolites, and extracellular vesicles, cultivating a persistent cycle of chronic neuroinflammation. This self-reinforcing process transcends individual neurons and involves critical components such as the blood-brain barrier and the neurovascular unit, providing insights into the shared patterns of injury progression observed in many neurodegenerative and cerebrovascular diseases.
Ultimately, this research calls for a paradigm shift in how we perceive glial cells, urging the scientific community to recognize them not merely as inflammatory agents but as essential regulators that integrate metabolism, immunity, and the stability of neurovascular systems. This new understanding could pave the way for innovative therapeutic strategies that aim to reprogram glial states and restore balance within the brain.
Read These Next

Eye-Scanning Tech: Verifying Humanity in Digital Interactions
Exploring the introduction of eye-scanning technology by Tinder and Zoom for user verification and its implications in a digital world rife with AI impersonation.

Can a Digital Twin Create a 'Superworker'?
This article explores the potential of digital twins, like Richard Skellett's AI counterpart, to transform ordinary workers into 'superworkers' by enhancing their productivity and addressing key considerations around governance and ethical use.

Quantum Computing: Can Europe Lead the Tech Race?
An overview of the potential for Europe, particularly France, to lead in the quantum computing race, highlighting key players and their innovative approaches.
