China Dominates Global Brain Computer Interface BCI Market
China has made a significant advancement in the field of medical technology by approving the world's first invasive brain-computer interface (BCI) device specifically designed for individuals with tetraplegia. This groundbreaking device enables enhanced control over hand movements, marking a pivotal transition from clinical trials to real-world application.
The approval, granted by China’s National Medical Products Administration on March 13, allows the implantable BCI to be marketed. Developed jointly by Neuracle Medical Technology, based in Shanghai, and Tsinghua University, this innovative product represents a major milestone not only in the country but also in the global BCI landscape.
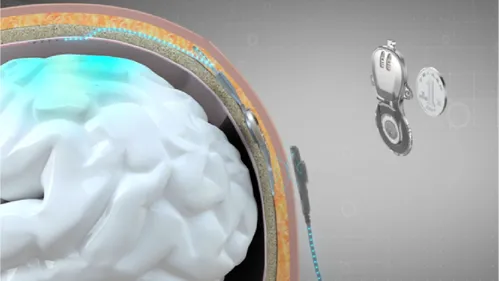
Brain-computer interfaces facilitate a direct line of communication between brain activity and external devices. By capturing signals generated in the motor regions of the brain through implanted electrodes, the system decodes these signals into actionable commands that can operate assistive technologies, greatly benefiting those with motor impairments.
This newly approved system targets patients with tetraplegia resulting from cervical spinal cord injuries, enabling them to perform simple hand functions through a pneumatic glove that responds to neural signals. This innovation is expected to considerably improve the quality of life for these patients.
The device comprises multiple components, including a BCI implant, an implanted electrode array, a neural signal transmitter and receiver, pneumatic glove equipment, and software for brain-signal decoding. This comprehensive system allows for more effective interactions between the user’s neural activities and the assistive technology.
The minimally invasive nature of the surgical procedure—where electrodes are placed between the skull and the dura mater—enables quicker recovery times, allowing patients to return home approximately one month after the surgery.
Following the device's approval, the National Healthcare Security Administration moved swiftly to review technical specifications and finalize medical insurance coding for the device. Consequently, this initiative aims to expedite the transition from approval to clinical utilization, ensuring that patients gain timely access to this essential technology.
In a competitive global landscape, various approaches to BCIs are emerging. Notably, in the United States, Elon Musk’s Neuralink is advancing fully implantable systems and has announced plans for its first human implantation in 2024, pending regulatory approval.
In Shanghai, StairMed Technology is developing fully invasive BCI systems and has recently completed strategic financing supported by major tech firms, including Tencent and Alibaba. Simultaneously, NeuroXess is investing in extensive manufacturing capabilities for implantable BCIs in China.
Data from CCID Consulting estimate that the BCI market in China is set to grow at an average annual rate of about 20 percent from 2024 to 2028, outpacing many traditional manufacturing sectors. By 2040, the domestic market could exceed 120 billion yuan, highlighting the country's burgeoning presence in this technological frontier.
China’s policies reflect ongoing support for the BCI sector, recognized as one of six key future industries in the national 15th Five-Year Plan. The approval of the first implantable BCI product not only marks a significant achievement for China but also sets a precedent for advancements in the global field of neuroscience and technology.
Ming Dong, vice president of Tianjin University, underscored that China has entered the first tier of global BCI development, citing technological advancements and increasing commercialization efforts. He noted that the country has achieved parity with international standards in non-invasive technologies, while closing the technological gap in invasive systems.
Read These Next

China Releases Guidelines for Global Sustainable Development Cooperation
The NSFC issued guidelines for the SDIC to tackle global issues and enhance international research collaboration.

China targets 80 years average life expectancy by 2030
China aims to boost average life expectancy to 80 years by 2030, focusing on healthcare enhancements and disease prevention.

China Emerges as Leader in Global Scientific Publishing
At the Zhongguancun Forum in Beijing, Arnold Pippel noted China's lead in scientific publications and praised AI's role in research integrity.
